Realisation of Precision Medicine
Cancer Genome
Cancer Risk Prediction Test
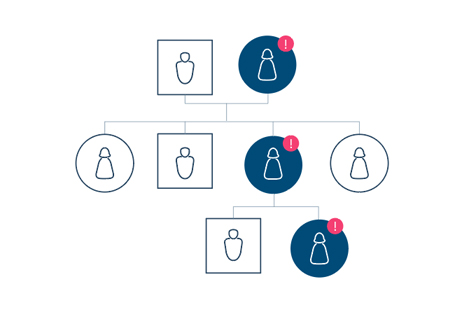
Cancers have various causes, but some cancers are caused by genetic mutations of specific inherited genes. People who have such cancer-related mutations have far more risk of cancer than others. Hereditary cancers are caused by gene abnormalities inherited from parents, and the incidence of cancer in the same family is high. Thus, early detection and identification of the exact cause through gene diagnosis is required.
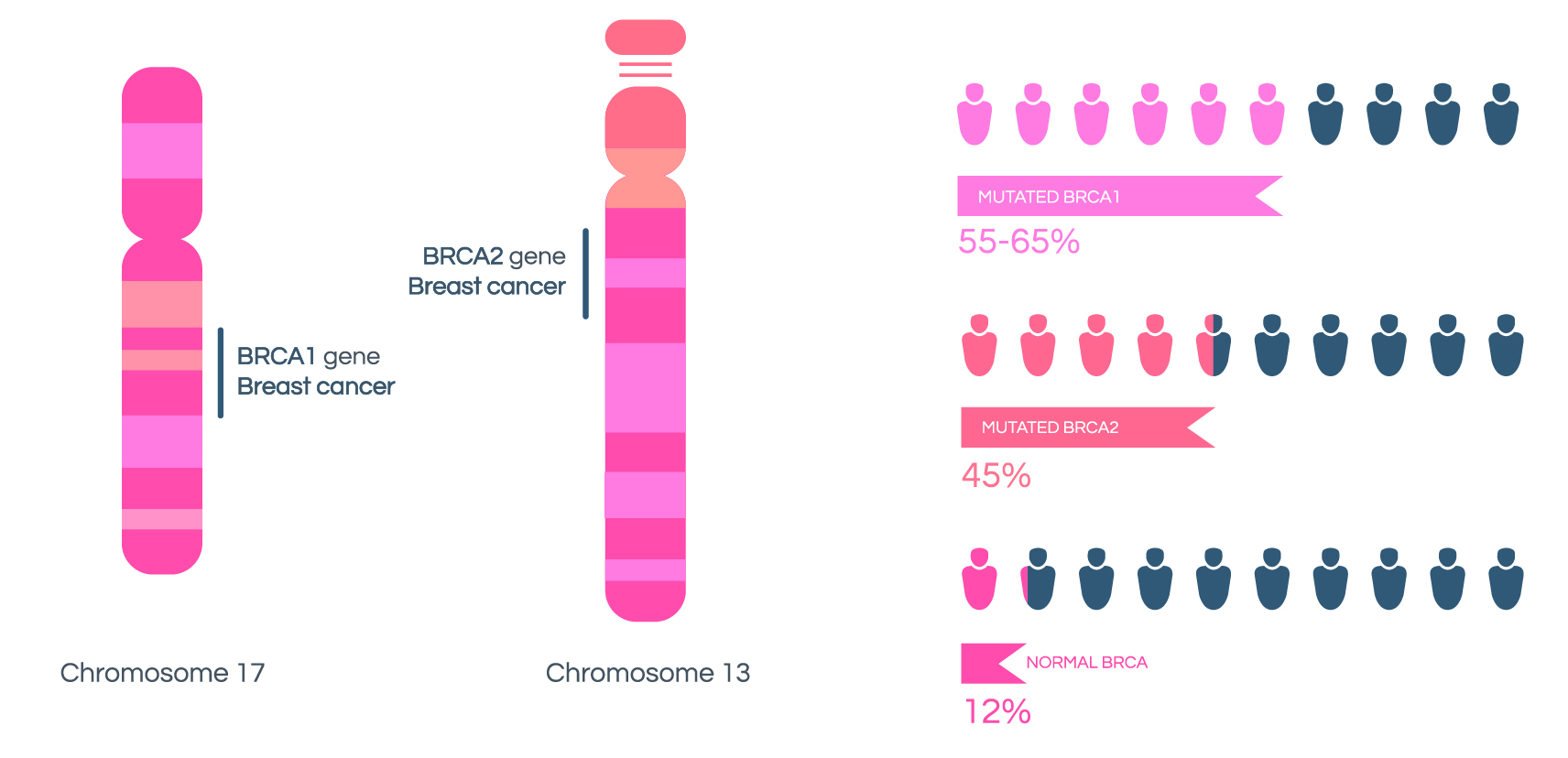
The most commonly known hereditary cancers are breast cancer, ovarian cancer, and colon cancer. For the incidence of breast cancer, 5%–10% is known to be genetically caused, and more than half of such cases are caused by abnormalities in genes called BRCA1 and BRCA2. When there is a mutation in the BRCA1 gene, the risk increases up to 87% after age 70 compared to when there is no variation. Moreover, various genes including MLH1 are found to be the main cause of colon/rectal cancer. With mutation, the incidence of cancer increases greatly.
Hereditary Cancer Panel
The Heredity Cancer Panel diagnoses the hereditary risk of cancer by analysing the causative gene related to hereditary cancer. Mutations of a total of 16 genes including BRCA1/2 genes, and genes related to Lynch syndrome, which is the main cause of colon cancer, are analysed.
| 9Clinical Diagnostics | Target | Description (related cancer type) |
| APC | Large Intestine | Germline mutations associated with familial adenomatous polyposis (FAP) |
| MUTYH | ||
| BRCA1 | Breast Ovary |
Related to breast/ovarian cancer |
| BRCA2 | ||
| CDH1 | Stomach | Responsible for cell-to-cell adhesion llmplicated in cancer progression and metastasis |
| Large Intestine | ||
| EPCAM | Large Intestine Rectum |
Related to the mismatch repair of DNA Lynch syndrome (hereditary non-polyposis colorectal cancer, HNPCC) |
| PMS2 | ||
| MLH1 | ||
| MSH2 | ||
| MSH6 | ||
| MEN1 | Endocrine System | Multiple endocrine neoplasia type 1 (MEN-1 syndrome) Familial neoplasia affecting the network of hormone-producing glands |
| PTEN | Thyroid Breast |
Cowden syndrome Tumor – suppressor gene |
| RB1 | Eye | Retinoblastoma |
| RET | Endocrine System Thyroid |
Proto-oncogene Multiple endocrine neoplasia type 2 Medullary thyroid carcinoma |
| TP53 | Breast Bone Etc. |
Li – Fraumeni syndrome |
| VHL | Eye Kidny Central Nervous System |
Tumor – suppressor gene von Hippel – Lindau Syndrome |
BRCA 1/2 Gene Test
All areas of the BRCA1/2 gene, which is the representative cause of hereditary breast cancer, are analysed to diagnose a person’s risk of breast cancer. Mutations in the BRCA 1/2 gene greatly increase the risk of breast cancer.
Cancer Cause Discovery Test
The Cancer Cause Discovery Test tracks the cause of cancer and provides important information for personalised cancer treatment by analysing various mutations of major genes involved in the occurrence of cancer, using DNA extracted from the tumour samples of a patient. Cancers used to be classified in a histopathological manner, but now gene analysis is performed on cancer tissue and cancers are divided into detailed items according to the gene mutation, which contributes to improved response (about 75% of patients exhibit different effects when the same anticancer drug is prescribed) and provides personalised treatment for cancer patients while minimising adverse effects.
Regular Cancer Monitoring Test
When cancer occurs in the body, NDA fragments detach from cancer cells in the blood. This is called ctDNA (circulating tumour DNA). When ctDNA is checked regularly, cancer cells can be detected early and prompt measures taken.
Unlike existing biopsies, this cancer-monitoring test can be performed by merely collecting blood, which makes regular check-ups more efficient. However, the test is not widely given because the levels of ctDNA are different by type of cancer and individual, and detection is very difficult. Macrogen is expanding the service by continuously making technical improvements.
Prenatal Genome
Non-Invasive Prenatal Screening (NIPS)
NIPS is the test that screens for aneuploidy of chromosomes in a foetus using its DNA (cfDNA). The test is performed on Down syndrome, Edwards syndrome and Patau syndrome, the most typical kinds of aneuploidy, and aneuploidy of gender chromosomes.
NIPS is safer and allows checking for abnormalities of the foetus earlier than the existing invasive amniotic fluid testing. Pregnant women aged 35 or older or those with a family history of foetal abnormalities should take the test because the risk of chromosome aneuploidy is very high.
Rare Disease Genome
Rare Disease Genome
There are more than 8,000 known rare diseases. Among them, 80% are hereditary diseases caused by gene mutation. Even though the prevalence rate of each disease is very low, the risk is rather high around the world because there are so many types and symptoms of these diseases.
- 30% of patients with rare diseases die before they reach the age of 5
- 50% of patients with rare diseases are children
- 80% of patients with rare diseases are willing to actively participate in studies
- On average, patients with rare diseases receive incorrect diagnoses about six times, and the correct diagnosis is usually made after ten years of evaluations.
In the United States, lifetime medical costs for one patient with a rare disease reach about $5 million
Rare Genetic Disease Screening
Although many rare diseases are known, different degrees of symptoms can be shown for the same disease and there are multiple diseases with similar symptoms. Thus, it is still very difficult to receive a correct diagnosis and treatment. Rare genetic disease screening is for patients with obscure symptoms that need a correct diagnosis and testing to establish a guideline for appropriate therapy.
New personalised drugs are being developed, although there are not many drugs for rare genetic diseases. Thus, the need for a correct diagnosis is rising.
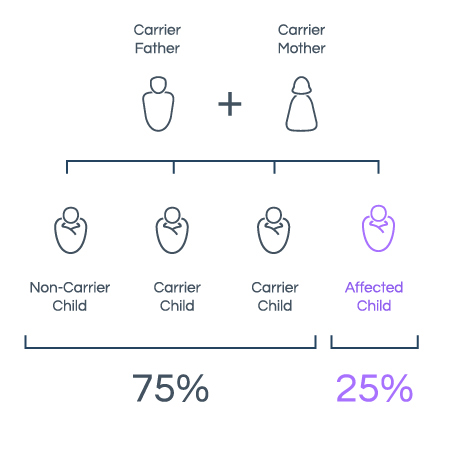
Carrier Screening
Carrier screening is a test to aid healthy family planning by predicting the genetic risk to future children by analysing the parents’ genomes.
In the case of autosomal recessive diseases, if the parents are carriers who have genetic risk factors, even though they appear normal, their children have a 25% possibility of being born with a hereditary genetic disease.
NBS (Newborn Screening)
NBS is a genetic disease screening test performed on newborns within 24–48 hours of birth. The purpose of the test is to prevent the risk of children’s hereditary genetic diseases by testing for metabolic disorders, which are dangerous diseases that occur within 1 year of birth but can be treated when detected early.